 
|
|
Welcome at JVSE.de |
About JVSE |
Consulting & Engineering |
Production with CO2-technologies |
Imprint |
Contact |
What is supercritical CO2?
|
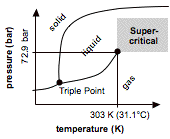
Materials can generally exist in three states: solid, liquid and gas. It is possible to can change the state from one to another by altering temperature and/or pressure. The diagram illustrates the conditions under which carbon dioxide will be solid, liquid or gas. However, if temperature AND pressure are both increased starting at the "critical point", the distinction between liquid and gas eventually disappears. Above this, carbon dioxide behaves partly like liquid, and partly like gas. It spreads out like a gas to fill the available space. At the same time supercritical carbon dioxide can dissolve substances as if it were a liquid. Carbon dioxide, becomes supercritical above 72.9 atmospheres of pressure and 31.1 degrees Centigrade. Compared to other substances, both temperature and pressure of supercritical carbon dioxide are relatively mild. The low temperature makes supercritical carbon dioxide particularly convenient, since it can be used with substances that would otherwise be damaged by the high temperatures required by conventional solvents. One of the first large-scale applications of supercritical carbon dioxide was to remove caffeine from tea and coffee. This process is now used in preference to the original solvent, methylene chloride, a suspected carcinogen. Other applications of supercritical carbon dioxide include the extraction of hops, pharmaceuticals, essential oils, and other temperatur sensitive, hardly soluble. New applications of supercritical carbon dioxide include particle generation (e.g. RES-process ...) and micro-encapsulation (e.g. CPF, PGSS ...). JVSE develops, produces and markets novel products that are based on high-pressure and sc CO2-technologies, respectively. Key markets for our products are are the cosmetics, detergent, chemical and food industry. We realise your ideas. Please do not hesitate to contact us. | ||
|
Copyright (c) 2006 Joachim von Schnitzler
We are pleased to offer you: Consulting, Engineering and toll production with CO2-technologies (supercritical carbon dioxide), e.g. particle generation (nano- and micro-scale) and micro encapsulation. | |||